
Recently we received some inquiries like “Even though it was made of stainless steel, it soon got rusted” from our customers.
Stainless steel is a very rust-resistant alloy of iron.
Indeed, compared to iron, it is quite hard to rust, but it doesn't mean it doesn't rust at all.
Why is stainless steel so resistant to rust?
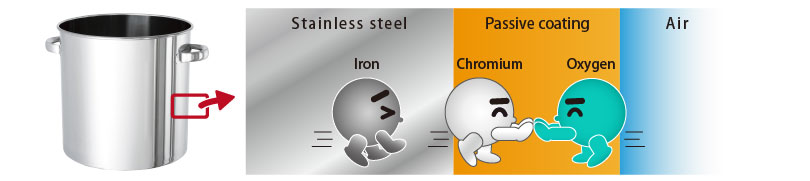
When exposed to air, it will form a self-healing barrier!
When exposed to air, it will form a self-healing barrier!In stainless steel, chromium bonds with oxygen in the air (=oxidation) before iron, which causes rust, and forms a very thin passive film (protective coating) of a few nanometers.
The passive film is resistant to chemical change and it is so strong that can prevent iron from oxidation(=rust).
Passive coating can be damaged and torn, but it can self-heal in a moment, so it does not leave room for iron to rust.
Our company ‘s standard material, SS304, is made of 18% chromium, 8% nickel and iron.
(Also called “18 -8 Stainless Steel”, 18 and 8 refer to the ingredient.)
Nickel helps make passive coatings easier to form.
"It should be hard to rust, but it still got rusty." Why?
"Contagious" corrosion
If a stainless-steel container is left with a metal such as iron on the surface, when the metal was rusted, the stainless container itself will go rust, too.
Corrosion occurs not only between stainless steels but also in contact with metals other than stainless steel.

Contaminated with Dirt or Moisture
If dirt or moisture remains on the surface of the stainless steel, the passive film cannot be formed on that part, and the stainless steel will become easy to be rust.

When Contacted with Salt
Compared to the other metals, stainless steel is much more resistant to salt.
But if left the salt attached to the surface, it still will go rust.
Although it depends on the material, but generally stainless-steel containers are not suitable for storing contents with salt.

Acids and Alkalis content
Acids and Alkalis contentAlthough it depends on the kind of acid or alkali, those substances really don't go well with stainless steel, and eventually the container will be rust.
Also, the effect changes depending on the concentration and temperature of chemicals.


Rust-Resistant Stainless Steel Container
This container is made of a rust-resistant stainless material called SUS 316 L. SUS 316 L is more rust-resistant than standard SUS 304 stainless steel. These containers are ideal for highly hygienic production sites such as pharmaceutical factories, cosmetic factories and semiconductor factories.
Which part of stainless-steel containers rust easily?
Places where Water and Dirt tend to Accumulate
The corners of the container, the back of the handle, the edge part, and other parts that are hard to wash and easy to get dirty, and the moisture tends to accumulate in there.
So that those part are rust easily.

Damaged part
The dirt and moisture in the wound of the container may cause rust.
And if the damage is made by another metal-made substance, the possibility of the container catching the “contagious corrosion” will be increased.

Adjacent to the Base or Floor
The bottom is easy to be scratched because it contacts with the base or the floor.
If the place where you put it is dirty, it will be contaminated.
If you leave the container on the metal while it is wet, it will cause corrosion.

The Machined Part
The moisture and dirt is prone to remained at the parts which are made of by pressing or welding.
The surface composition may be changed, so that it rusts easily.


Rust-Resistant Stainless Steel Container
This container is made of a rust-resistant stainless material called SUS 316 L. SUS 316 L is more rust-resistant than standard SUS 304 stainless steel. These containers are ideal for highly hygienic production sites such as pharmaceutical factories, cosmetic factories and semiconductor factories.
Tag Related Posts
- No related posts.
